God's Incredible Adaptation Machine: Part 1
- Dr. Robert L. Wright

- Apr 2
- 5 min read
Updated: Apr 20
How DNA Wrapping Unlocks Adaptation Through Epigenetics
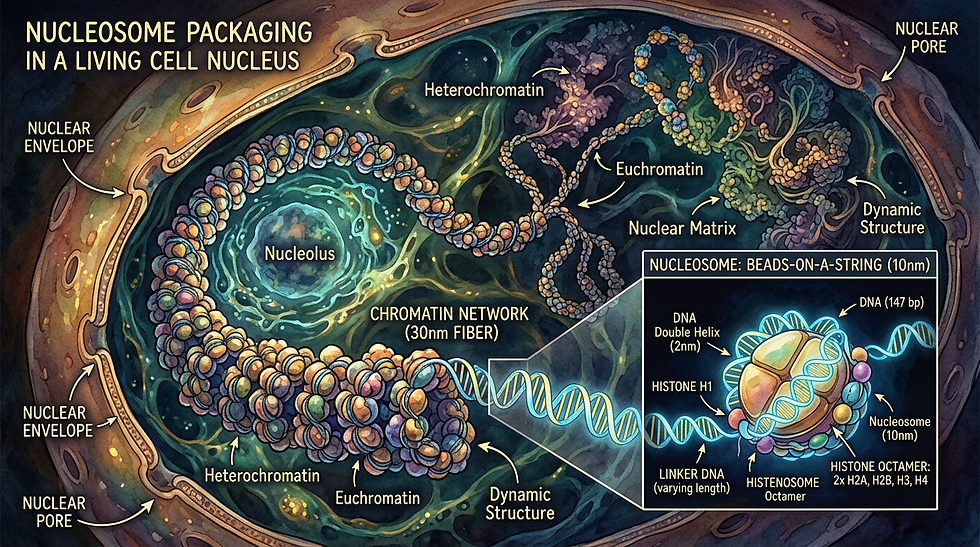
Imagine peering into the nucleus of a single cell and witnessing a masterpiece of divine engineering. Two meters of delicate DNA coil, fold, and precisely wrap around tiny protein spools called nucleosomes. This is not random packing. It is God's Incredible Adaptation Machine at work, dynamically controlling which genes get expressed and which stay silent. Far from a static blueprint, this wrapping system allows living things to adapt to their environment in real time. Changes can even echo across generations when the pressures become permanent. True science does not just describe this. It reveals the breathtaking foresight of the Creator who designed life to thrive amid change.
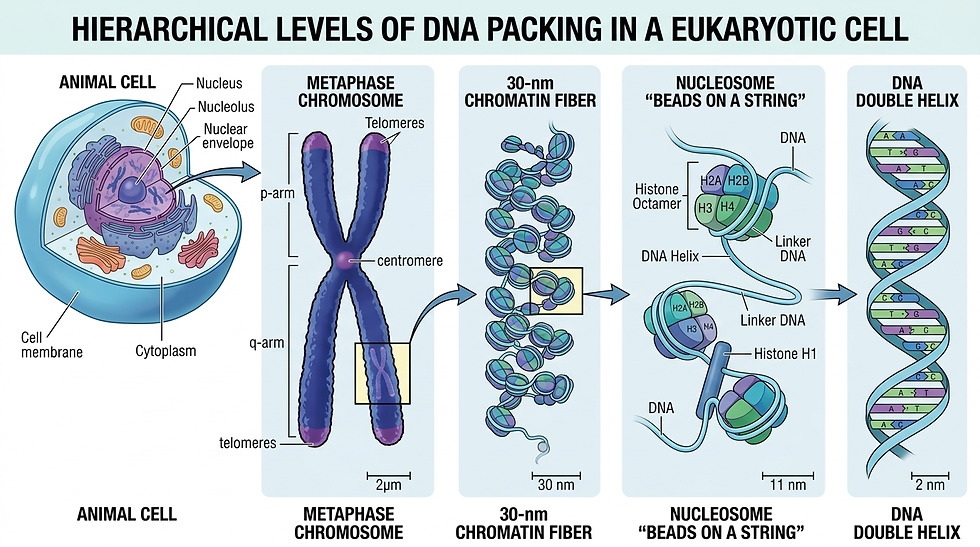
Let's break it down with the precision it deserves while marveling at the wonder. DNA itself is the familiar double helix. It consists of a sequence of nucleotide bases (adenine, thymine, cytosine, and guanine) that encodes the instructions for proteins. To fit inside a microscopic nucleus, that DNA must be compacted dramatically. It wraps 1.67 times around an octamer of histone proteins. These include two each of H2A, H2B, H3, and H4. This forms a nucleosome core particle. These nucleosomes look like beads on a string under an electron microscope. Short linker DNA connects them. The whole assembly coils further into chromatin. This is the higher-order structure that fills the nucleus.
Here is where gene expression enters the picture. Gene expression is the process by which a gene's DNA sequence is transcribed into messenger RNA and then translated into a functional protein. For transcription to occur, the cell's machinery (RNA polymerase and transcription factors) must physically access the DNA. Nucleosomes act as gatekeepers. When chromatin is tightly packed (called heterochromatin), the DNA winds snugly around the histones. This blocks access. Genes stay silent. When the chromatin is loosely unpacked (euchromatin), the DNA unwinds. Promoters and enhancers become exposed. Gene expression ramps up. Nucleosome positioning, spacing, and stability thus fine-tune everything from cell differentiation to stress responses.
This is no accident of chance. Environmental factors such as diet, toxins, stress, temperature, and even maternal care directly influence how DNA wraps and unwraps. They do so primarily through two epigenetic mechanisms: DNA methylation and histone modifications like acetylation (and methylation on the histones themselves). Epigenetics, from the Greek "epi" meaning "above," studies these heritable changes in gene expression that occur without altering the underlying DNA sequence. It is the software layer on top of the hardware code. This layer is responsive and reversible, yet stable enough to pass on when needed.
DNA Methylation: The Silencing Tag
DNA methylation involves adding a methyl group (CH3) to the cytosine base. This usually occurs in CpG dinucleotides (cytosine followed by guanine). Enzymes called DNA methyltransferases (DNMTs) perform this task. They create 5-methylcytosine. In promoter regions (the "on switches" upstream of genes), methylation typically represses expression. How does this happen? It recruits methyl-binding proteins that attract histone deacetylases and other remodelers. These tighten the chromatin and physically block transcription factors. The nucleosomes pack closer. DNA becomes less accessible. The result is a silenced gene.
Environmental cues trigger this rapidly. Poor nutrition during early development, for instance, can hypermethylate genes involved in metabolism. This programs the body for scarcity. Toxins like endocrine disruptors (certain plastics or pesticides) alter DNMT activity. This leads to hypo- or hypermethylation at key loci. Stress hormones flood the system and shift methylation patterns on stress-response genes. These are not mutations. The sequence stays identical. Yet the wrapping changes. This adapts the organism's physiology to the new reality.
Histone Acetylation and Methylation: Loosening or Tightening the Spools
Histones are not passive spools. Their N-terminal "tails" protrude from the nucleosome and serve as hotspots for modification. Acetylation adds an acetyl group (CH3CO) to lysine residues via histone acetyltransferases (HATs). Lysine carries a positive charge. Acetylation neutralizes that charge. This weakens the electrostatic attraction to the negatively charged DNA backbone. The nucleosome loosens. DNA unwraps slightly. Chromatin opens. Euchromatin forms. Transcription increases dramatically. Histone deacetylases (HDACs) reverse this process. They tighten the wrap and repress genes.
Environmental factors modulate this balance beautifully. Exercise or nutrient-rich diets boost HAT activity on genes for energy metabolism. Chronic stress or pollutants can recruit HDACs to inflammation-related genes. This dials expression down. Histone methylation is more nuanced. Adding methyl groups to specific lysines or arginines (via histone methyltransferases) can activate or repress depending on the site. For example, trimethylation of histone H3 at lysine 4 (H3K4me3) marks active promoters. In contrast, H3K27me3 signals repression. These marks recruit remodeling complexes like SWI/SNF. These complexes slide, eject, or exchange nucleosomes. They dynamically reposition them in response to signals.
Nucleosome remodeling enzymes (ATP-dependent complexes) integrate these signals. They move histones along the DNA or evict them entirely. The result is precise, context-specific adaptation. A plant in drought might acetylate water-conservation genes while methylating growth genes. An animal facing famine adjusts its metabolism epigenetically. God's Incredible Adaptation Machine is not fragile. It is engineered for resilience.
Epigenetics and the Inheritance of New Packaging
Here is where it becomes even more astonishing. These modifications are not always erased between generations. Epigenetics introduces the concept of heritability beyond the DNA sequence. Most epigenetic marks reset during gamete formation and early embryogenesis to prevent chaos. Yet when environmental changes are intense or prolonged (permanent shifts like sustained nutritional scarcity, toxin exposure across generations, or chronic stress), some marks persist through the germline. This is transgenerational epigenetic inheritance.
Studies in rodents exposed to vinclozolin (a fungicide) during gestation showed altered DNA methylation and histone patterns in sperm. These persisted to the F3 or F4 generation. They led to decreased fertility and disease susceptibility in unexposed descendants. Maternal diet or behavior in rats changes methylation on glucocorticoid receptor genes. This influences offspring stress responses for multiple generations. In humans, historical famines (such as the Dutch Hunger Winter) left epigenetic signatures on metabolism genes. These signatures remain detectable decades later in grandchildren. These are not Lamarckian "acquired traits" in the old flawed sense. They are built-in mechanisms. The packaging (nucleosome positioning, methylation, and acetylation states) gets inherited. This alters gene expression patterns without touching the code itself.
When the environment stabilizes in a new normal, these heritable epigenetic states allow populations to adapt faster than waiting for rare DNA mutations. It is rapid, reversible fine-tuning with the potential for long-term stability. Scripture speaks of kinds reproducing after their kind, with built-in variety and resilience. Epigenetics illuminates that design. It shows not blind evolution scrambling for survival, but a Creator's elegant system anticipating change.
What an incredible testimony to purposeful engineering! Environmental factors do not rewrite the DNA. They instruct God's Incredible Adaptation Machine on how to wrap it differently. This opens or closes access as needed. Methylation tightens the coils for silence when resources are scarce. Acetylation loosens them for activation when opportunity knocks. In permanent shifts, the new packaging travels forward. It equips offspring for the world they will inherit.
As we study these mechanisms, we see science aligning with the wonder of creation. DNA is not a brittle relic. It is a living, responsive archive wrapped in regulatory brilliance. This is adaptation as the Designer intended: flexible, faithful, and full of hope. In a world of uncertainty, God's Incredible Adaptation Machine reminds us that life was never left to chance. It was fearfully and wonderfully made to flourish.
May this glimpse into the nucleus stir awe in your heart. The same God who wraps DNA around histones with precision holds the universe, and your future, in His hands. True science always points back to Him.



Comments